Studymodel empowers clinical operations leaders to optimise timelines and mitigate delivery risk during early planning. Robust, defensible, and transparent plans at governance = fewer problems during study delivery.
register for early access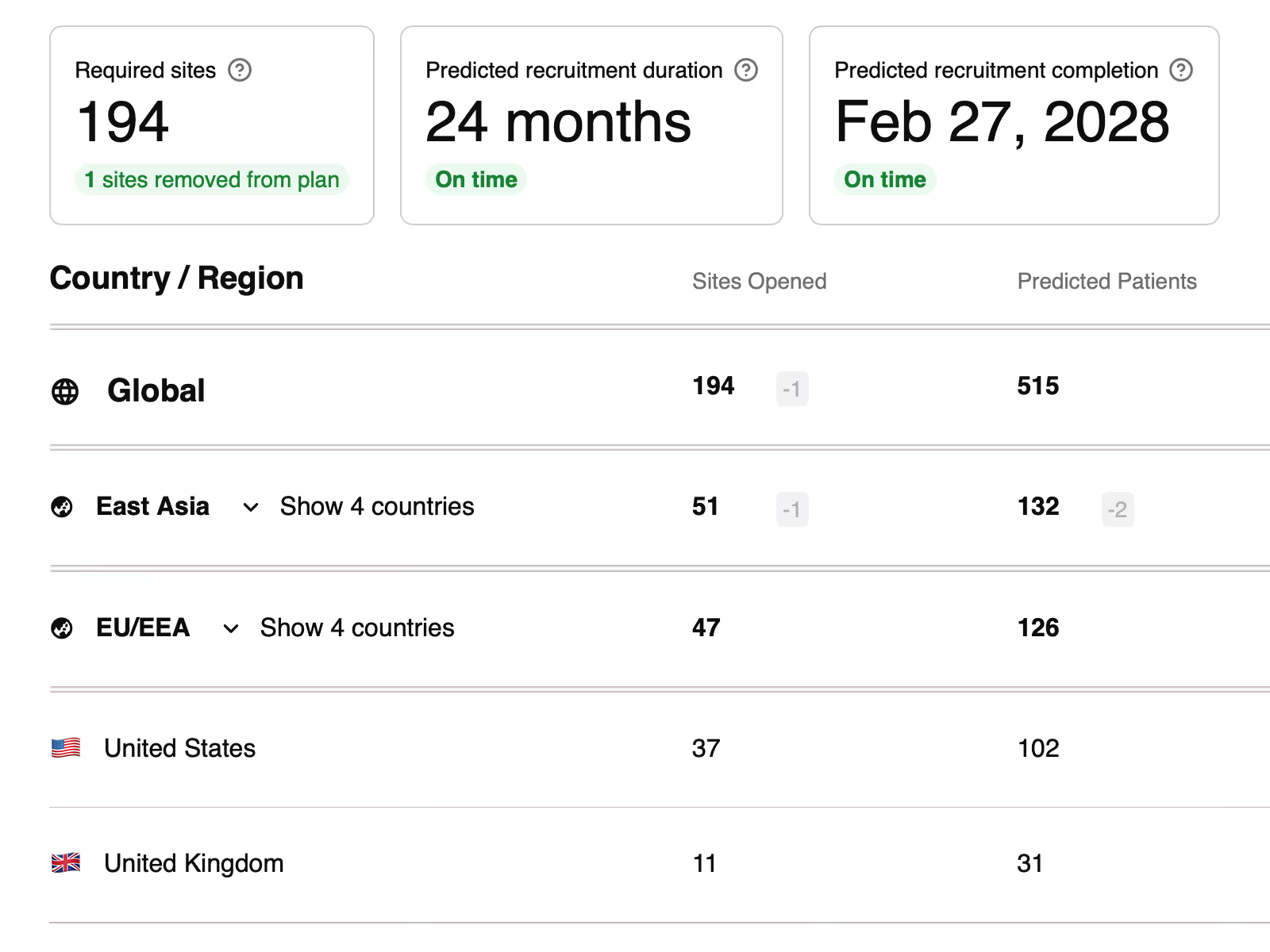
Stop wrestling with excel. We handle the math so you can focus on what matters.
video demo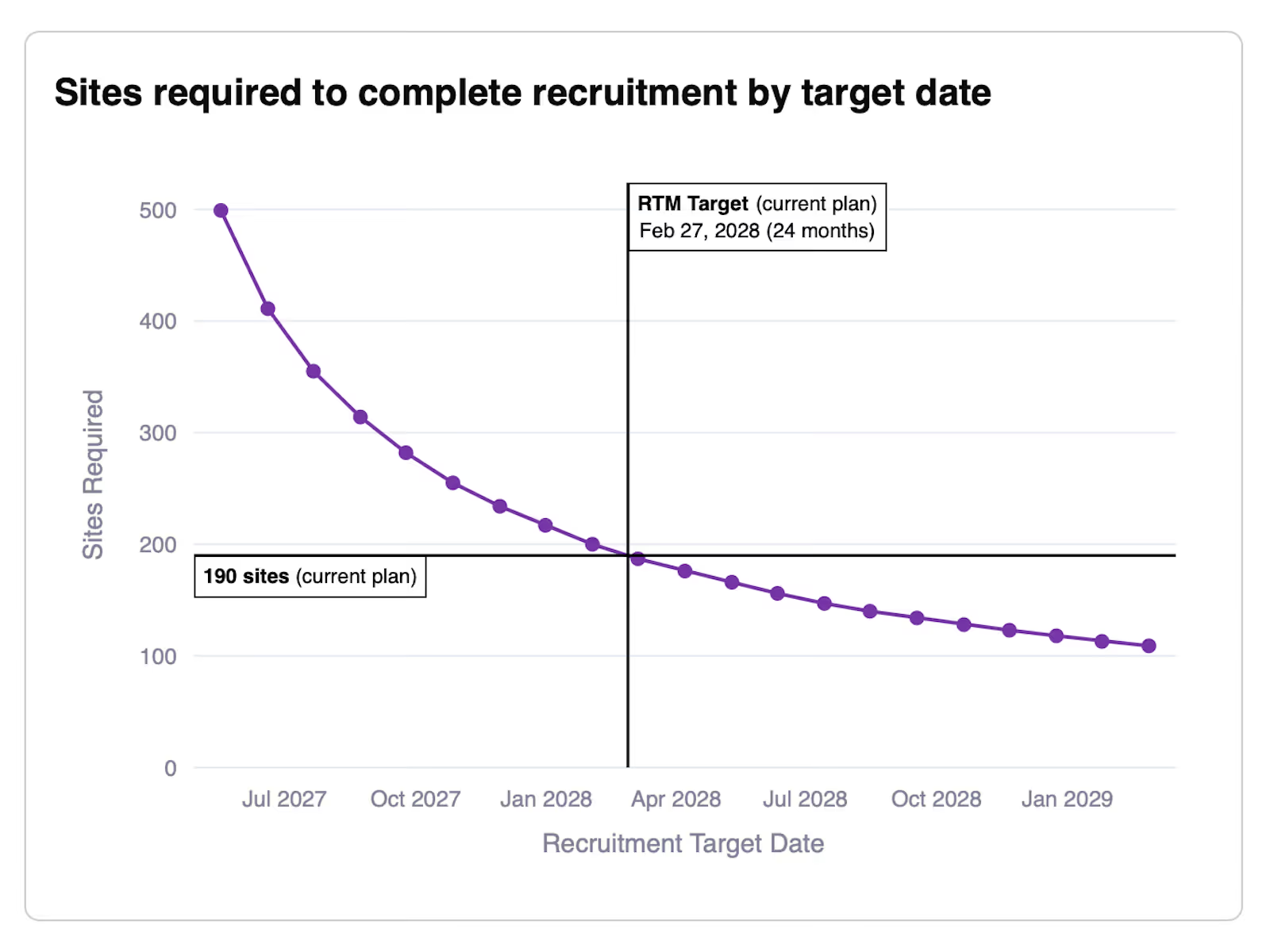
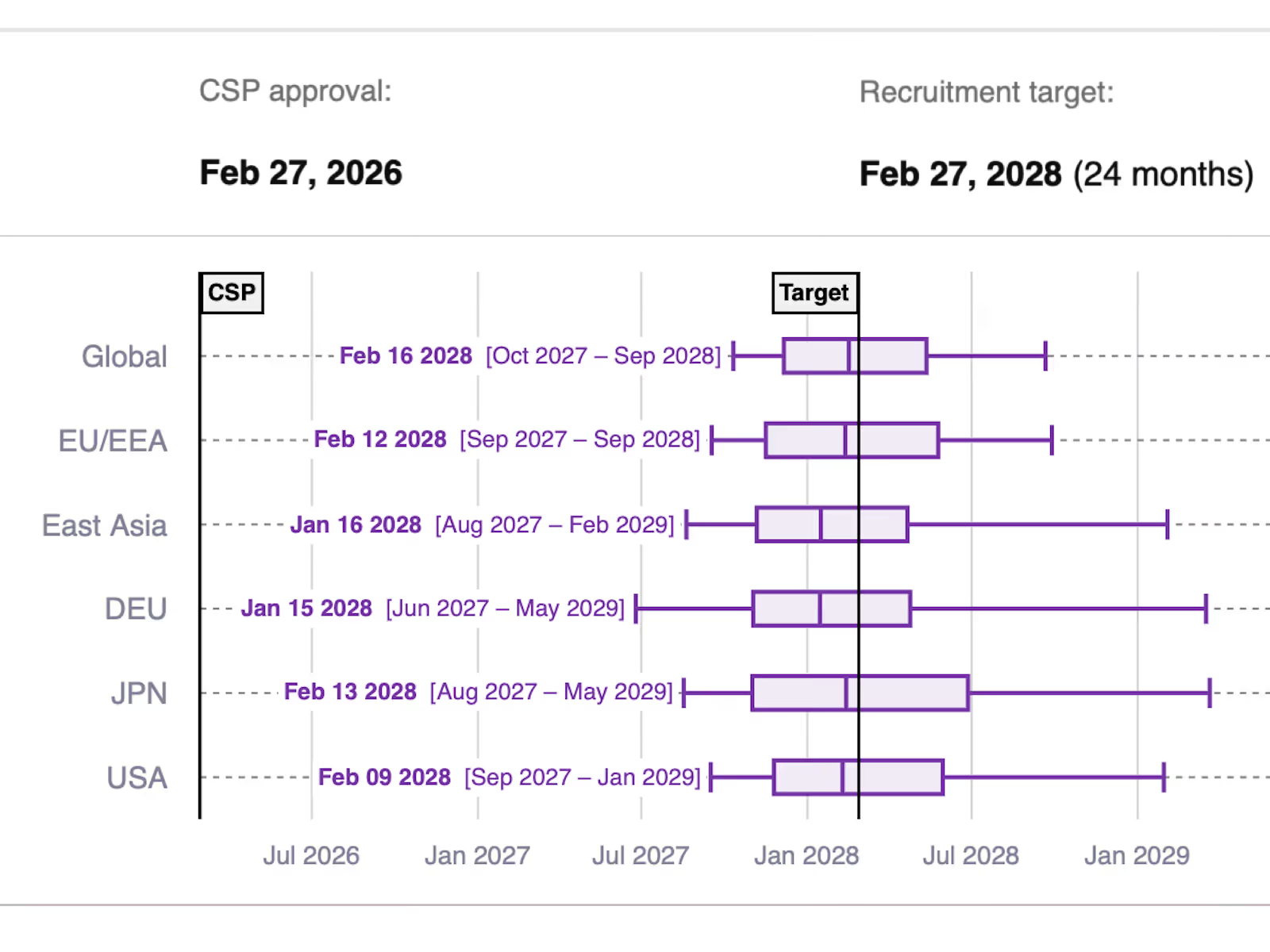
Identify risks and increase plan resilience by automatically simulating 1000’s of scenarios.
video demoBuild principled accrual estimates based on local prevalence and incidence data.
video demo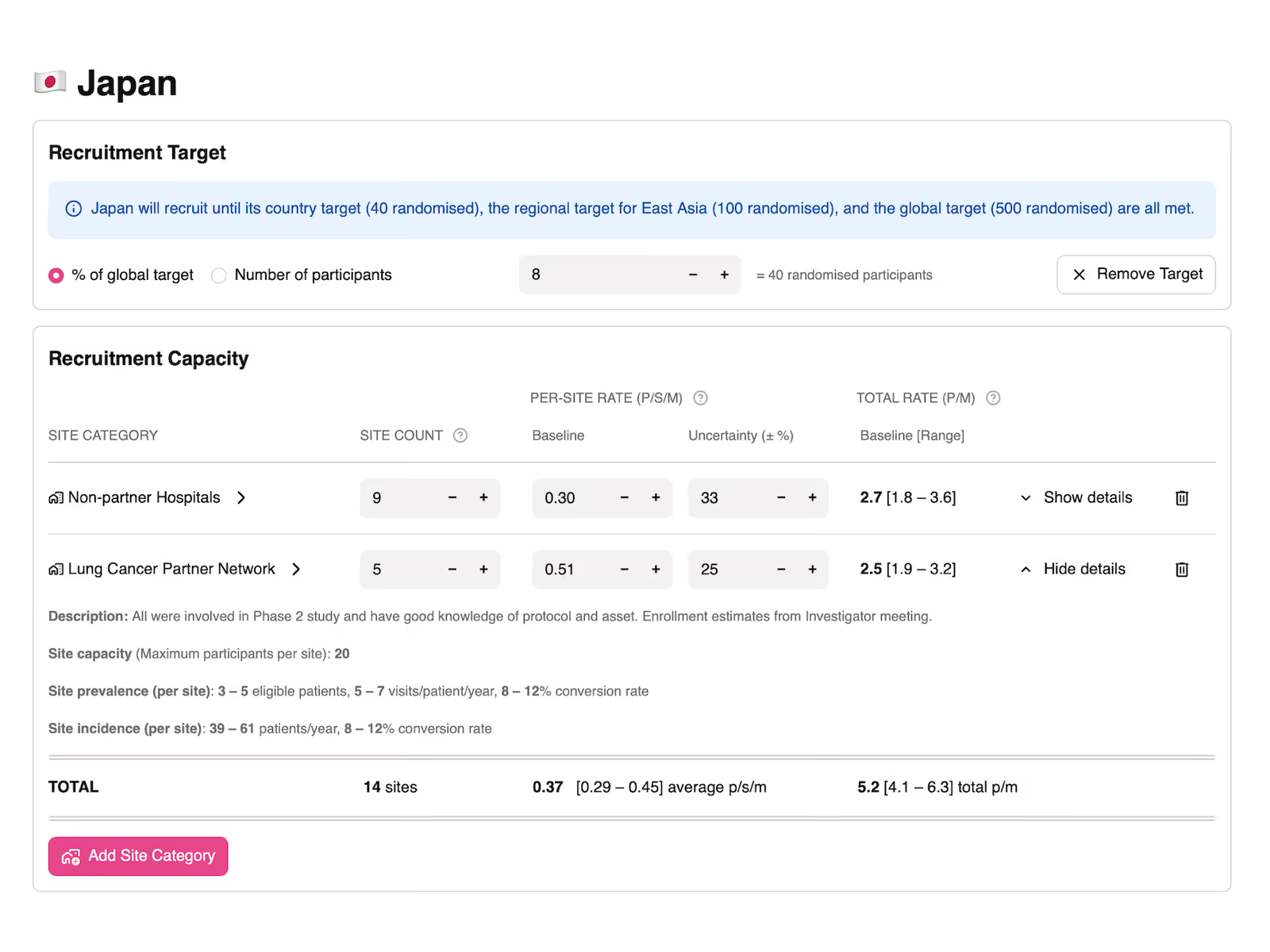
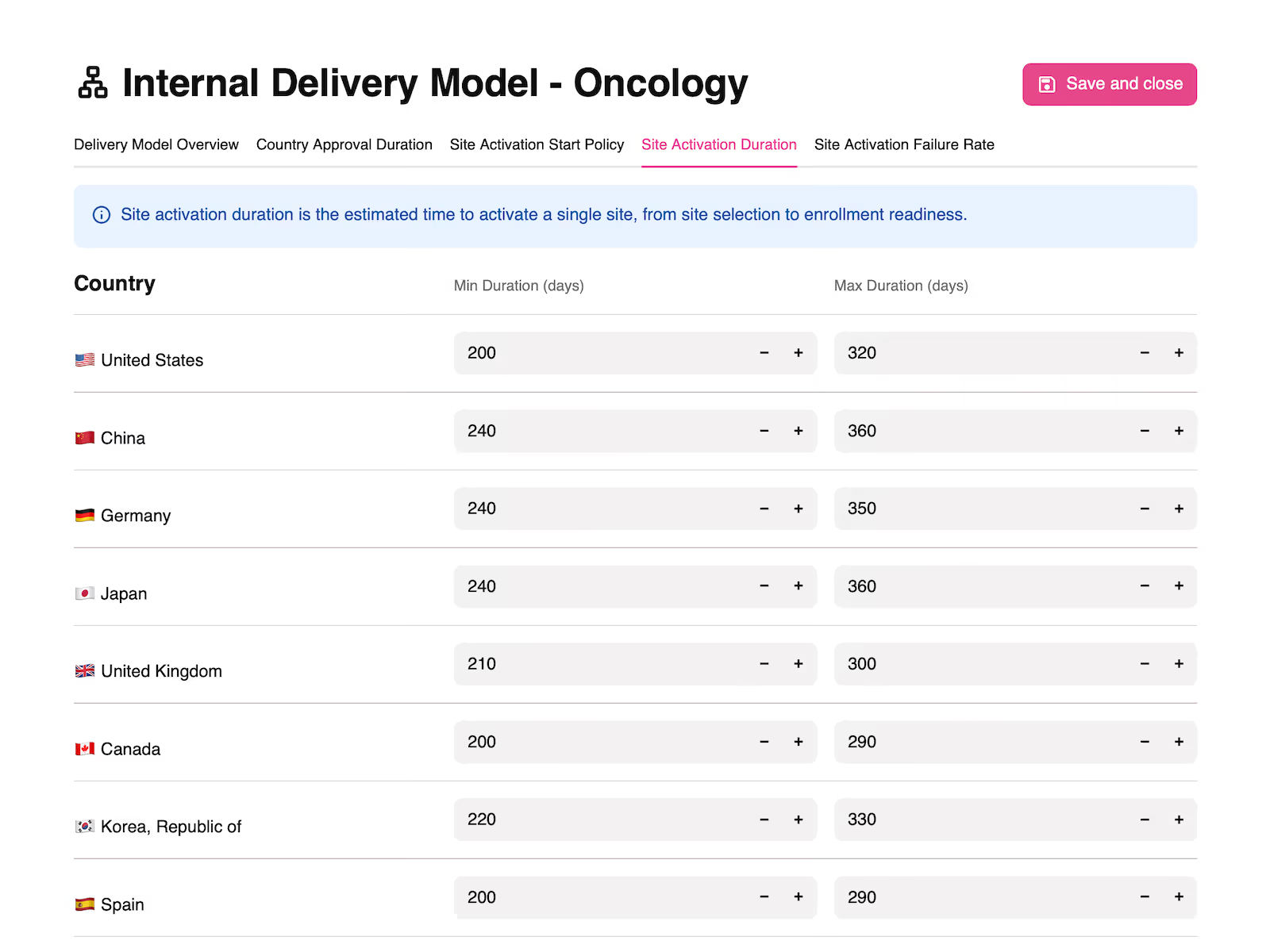
Embed expertise, data, and policy decisions in delivery model templates.
video demo